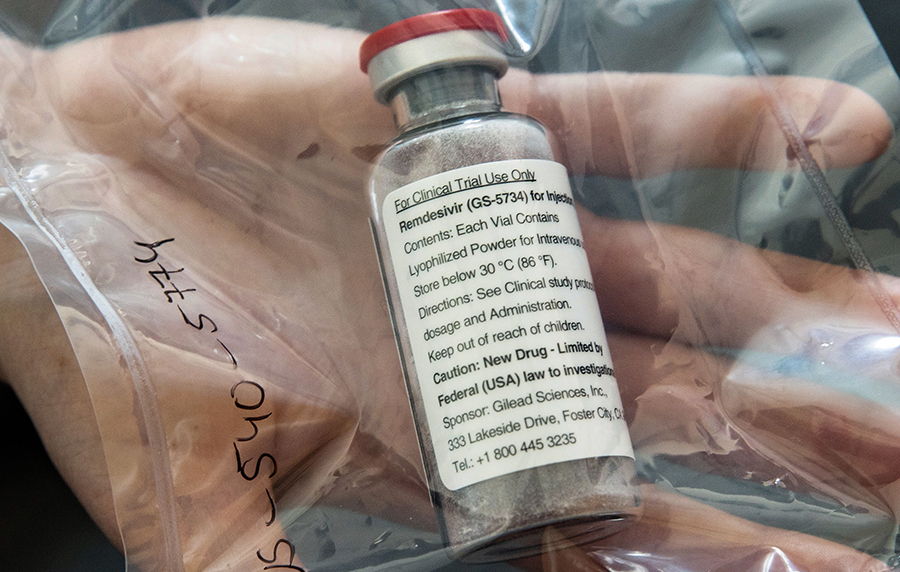
A World Health Organization (WHO) study has discovered that Gilead Sciences Inc’s remdesivir had little or no effect on COVID-19 patients’ length of stay in the hospital or chances of survival.
The only antiviral drug authorized for treatments of Covid-19 in the US failed to prevent deaths among patients.
The antiviral medication, among the first to be used as a treatment for COVID-19, was one of the drugs recently used to treat U.S. President Donald Trump’s coronavirus infection.
READ: AstraZeneca’s COVID-19 vaccine could be ready this year despite trial setback
The results are from WHO’s Solidarity trial, which evaluated the effects of 4 potential drug regimens, including remdesivir, hydroxychloroquine, anti-HIV drug combination lopinavir/ritonavir and interferon, in 11,266 adult patients across more than 30 countries.
The study found that the regimens appeared to have little or no effect on 28-day mortality or the length of the in-hospital course among patients hospitalized with COVID-19, the WHO said on Thursday.
READ: Dear Nigerians, the 5G Network is not a conspiracy theory
The results of the trial, which were posted online on Thursday, October 15, 2020, are yet to be peer-reviewed or published in a scientific journal
Earlier this month, data from a U.S. study of remdesivir by Gilead showed that the treatment cut COVID-19 recovery time by five days compared with patients who got a placebo in a trial comprising 1,062 patients.
Gilead told Reuters, “The emerging (WHO) data appears inconsistent, with more robust evidence from multiple randomized, controlled studies published in peer-reviewed journals validating the clinical benefit of remdesivir.
“We are concerned the data from this open-label global trial has not undergone the rigorous review required to allow for constructive scientific discussion, particularly given the limitations of the trial design.”
READ: Covid-19: WHO says not to expect first vaccination until early next year
Remdesivir, which was originally developed as a treatment for Ebola and Hepatitis C, interferes with the reproduction of viruses by jamming itself into new viral genes.
WHO chief scientist Soumya Swaminathan said on Wednesday that during the study, hydroxychloroquine and lopinavir/ritonavir were stopped in June after they proved ineffective, but other trials continued in more than 500 hospitals and 30 countries.
Swaminathan said, “We’re looking at what’s next. We’re looking at monoclonal anti-bodies, we’re looking at immunomodulators and some of the newer anti-viral drugs that have been developed in the last few months.”
READ: COVID-19: US FDA approves blood plasma treatment, 70,000 patients treated so far
Remdesivir received emergency use authorization from the U.S. Food and Drug Administration on May 1, and has since been authorized for use in several countries.
Gilead, however, disputed the conclusions of the W.H.O. study on Thursday, noting that a variety of drugs and drug combinations had been evaluated under a wide range of circumstances and that more rigorous studies had found a benefit.